Quiet heroism: Brazil's COVID-19 vaccine trial volunteers
"When the vaccine trial came, I joined immediately."
Sao Paulo, Brazil -- Story by Amanda Perobelli/Reuters
From doctors weary of seeing patients die to relatives who lost their loved ones, thousands of Brazilians, like nurse Jane Cristina Dias Alves, have volunteered for COVID vaccine trials in one of the world's worst-affected countries in the hope their quiet heroism will save lives.
Latin America's largest country has become a major testing ground for vaccines because of the scale of its outbreak, which has seen more than 7.3 million people infected and over 180,000 killed by the coronavirus.

Denise Abranches, 47, coordinator of dentistry at a Sao Paulo hospital, was the first volunteer outside Britain to receive the trial vaccine from AstraZeneca Plc.
"I witnessed many lonely deaths here: patients who could not say goodbye to their relatives; relatives who could not say goodbye to their loved ones. When the vaccine trial came, I joined immediately," she told Reuters.
"This global effort of volunteers in search of a vaccine will be remembered and will go down in history," she said. "The gesture of love from volunteers. That's how I'd like to be remembered."

I think we will remember this moment as a great moment of learning. A chance for humanity to learn, to be more humane, to live better, and for the government to take a closer look at its people. -Luiz Augusto Menegazzo
In addition to the AstraZeneca vaccine, Brazil has also hosted trials for Johnson & Johnson, Pfizer Inc. and partner BioNTech, and China's Sinovac Biotech Ltd.
While President Jair Bolsonaro, who has been widely condemned for his handling of the pandemic, has pledged not to take any coronavirus vaccine even if its approved by Brazil's health authorities, many volunteers are convinced inoculations are the only way to end the crisis.

I think when we look to this period, one legacy will be how everyone – the scientists, the health workers, everyone – fought to stop the loss of lives to COVID-19...Children will study this in school to understand the fight and difficulties we lived. -Keller de Martini
Some, such as Monica Aparecida Calazans, a Brazilian nurse who participated in the Sinovac trial, felt a personal obligation to contribute.

"My brother, who is 47 years old had COVID-19," she told Reuters. "If I don't take the vaccine, we will never know if it works or not. For him, but for everyone else too, I had the drive to take part in the trial."
The biomedical center running Sinovac's late-stage trials in Brazil, the Butantan Institute, said on Wednesday the vaccine met the 50% threshold for efficacy set by Brazilian health regulator Anvisa. It pledged to release more detailed information within two weeks as part of global trials.

I decided to take part in the trials because there was a light at the end of the tunnel for this new disease that scares all of us … I lose patients every day. It's desperate. It's terrifying. -Arianna Cavalcante
Bolsonaro, a right-wing nationalist who is an outspoken critic of China's influence in Brazil, has opposed Sinovac's vaccine for political reasons.
More broadly, he has sought to curtail lockdown measures and minimized the severity of the virus, actions that critics say have increased its spread and the death toll among Brazil's 210 million people.

I decided to take part in the trial because of my sense of commitment – not only my professional commitment as a nurse, whose essence is to take care, but also a personal commitment, a human commitment. -Sergio Aparecido Cleto
Amid the severe economic impact and a faltering political response, some volunteers said the opportunity to participate in the trials gave them a sense of regaining control of their own destiny.
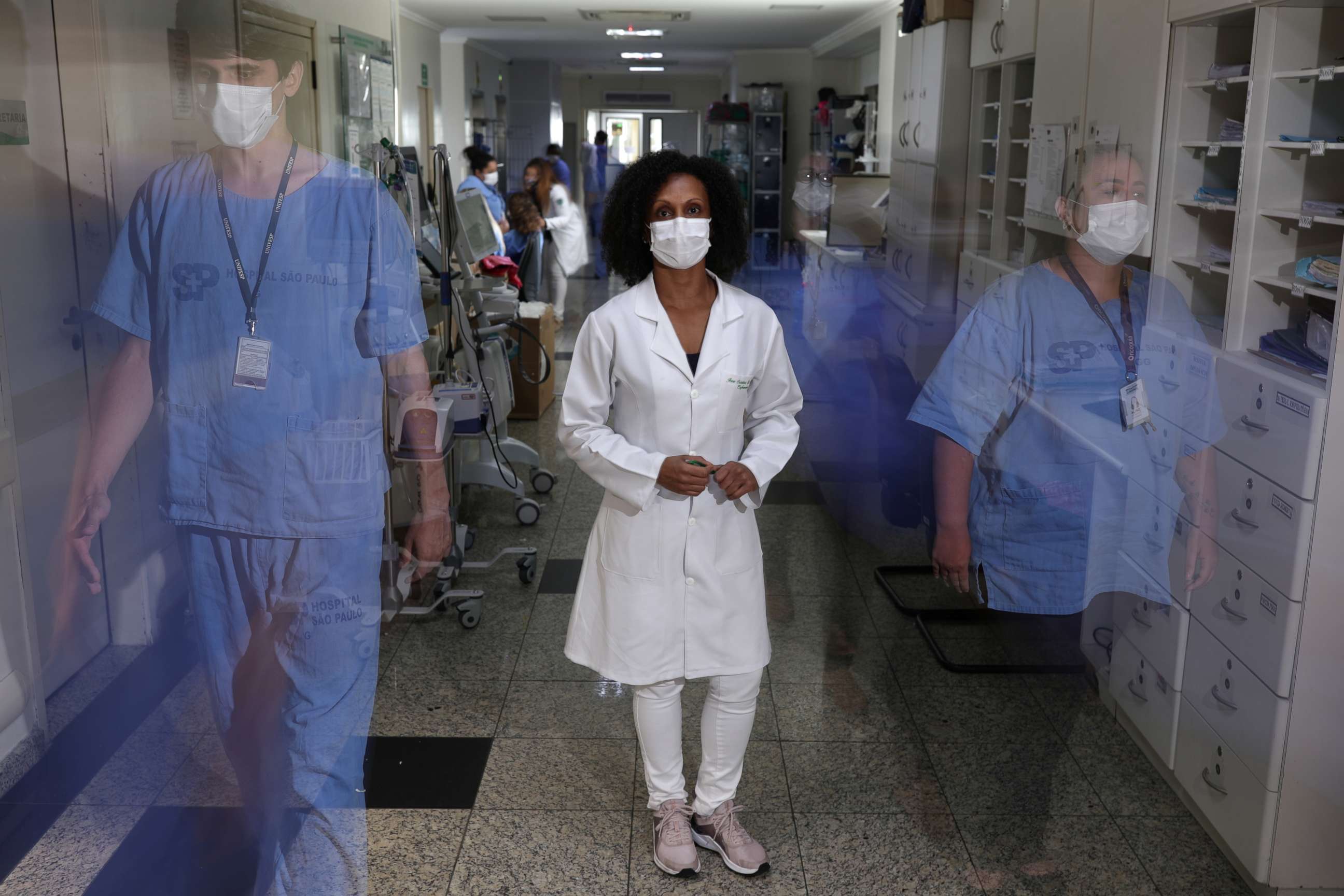
People look at me and say, 'Really, you are a volunteer? But you are so young. Isn't that risky?' And I try to put them at ease. We can also be an inspiration to people. To do this, to be a volunteer, it's important to understand your role in society. -Jane Cristina Dias Alves
"The hardest thing in the pandemic has been ... the sense of powerlessness that you can’t resolve the situation," said Ana Hial, a doctor who is also part of the AstraZeneca trial. "This global push to resolve the situation is a very special moment."
Trial volunteers do not usually know if they are receiving the actual vaccine or a placebo, meaning that even if the vaccines are ultimately successful, they may not have been protected from the disease all this time.
Some shrugged off what they said were mild side effects from the inoculation.

"The first days after I took (the vaccine), I had a few symptoms -- shivers and pain in my body," said Antonia Santos, a nurse who participated in the AstraZeneca trial. "My daughter was desperate. She said 'Mom, you are crazy.' I said, 'No, crazy would be not to take it.'"
Original Reuters Wider Image package credits: Photo Editing Marika Kochiashvili; Text Editing Dan Flynn and Lisa Shumaker; Layout Julia Dalrymple


