Pfizer COVID-19 vaccine now available for kids ages 5 to 11: 5 things to know
Pediatric vaccines are available at pharmacies, pediatricians' offices and more.
A COVID-19 vaccine is now available for kids ages 5 to 11, marking a major milestone in the nearly two-year coronavirus pandemic.
Dr. Rochelle Walensky, director of the Centers for Disease Control and Prevention (CDC), on Tuesday signed off on recommendations made earlier in the day by an independent CDC advisory panel, making the Pfizer vaccine available to the approximately 28 million children aged 5 to 11 in the United States.
"This is really just a time for parents to celebrate," Walensky said Wednesday on "Good Morning America." "We’ve done the due diligence, reviewed the science, made sure we have the appropriate doses for our children and really now we have parents who can have the peace of mind that when they get their kids vaccinated, they will be protected."
Walensky said parents with questions or concerns about vaccinating their child should speak with a trusted health care provider, adding, "we are here to answer your questions."
"We are now beginning to roll out, to distribute the vaccine, to give parents the information that they need," she said. "Go talk to your pediatrician, your trusted health care provider, your pharmacist and get the information that you need as we start scaling up vaccines across this country."
CDC director talks authorization of COVID vaccine for young children
Here are five things parents should know about COVID-19 vaccines and kids under the age of 12.
1. The vaccine will be distributed to kids through pediatricians, pharmacies, health clinics and more.
Shipping has already begun on the first batch of 15 million Pfizer pediatric vaccine doses after the White House purchased 65 million doses, more than enough to fully vaccinate all kids ages 5 to 11 in the U.S.
The vaccinations are expected to become widely available the week of Nov. 8. They will be accessible at pediatricians' offices, children's hospitals, pharmacies like CVS, Walgreens and Rite-Aid, and school and community-based clinics.

Parents can search for appointments at Vaccines.gov to find a local provider.
Once they receive their first shot, children will be given a vaccine card, just like adults and adolescents.
2. The Pfizer dose is different for kids under age 12.
Children have different immune systems than adults, so it should be reassuring for parents the Pfizer vaccine has been shown to be safe in kids.
Kids ages 5 to 11 will be given a 10-micrograms dose, one-third of the adolescent and adult dose.
Like with adults and adolescents, the pediatric vaccine will be delivered in two doses, three weeks apart.
A child will be considered fully immunized two weeks after their second dose.
To troubleshoot any confusion in the distribution of Pfizer's vaccine, orange-capped vials will contain doses for kids aged 5 to 11, while purple-capped vials will contain doses for adult and older adolescents.
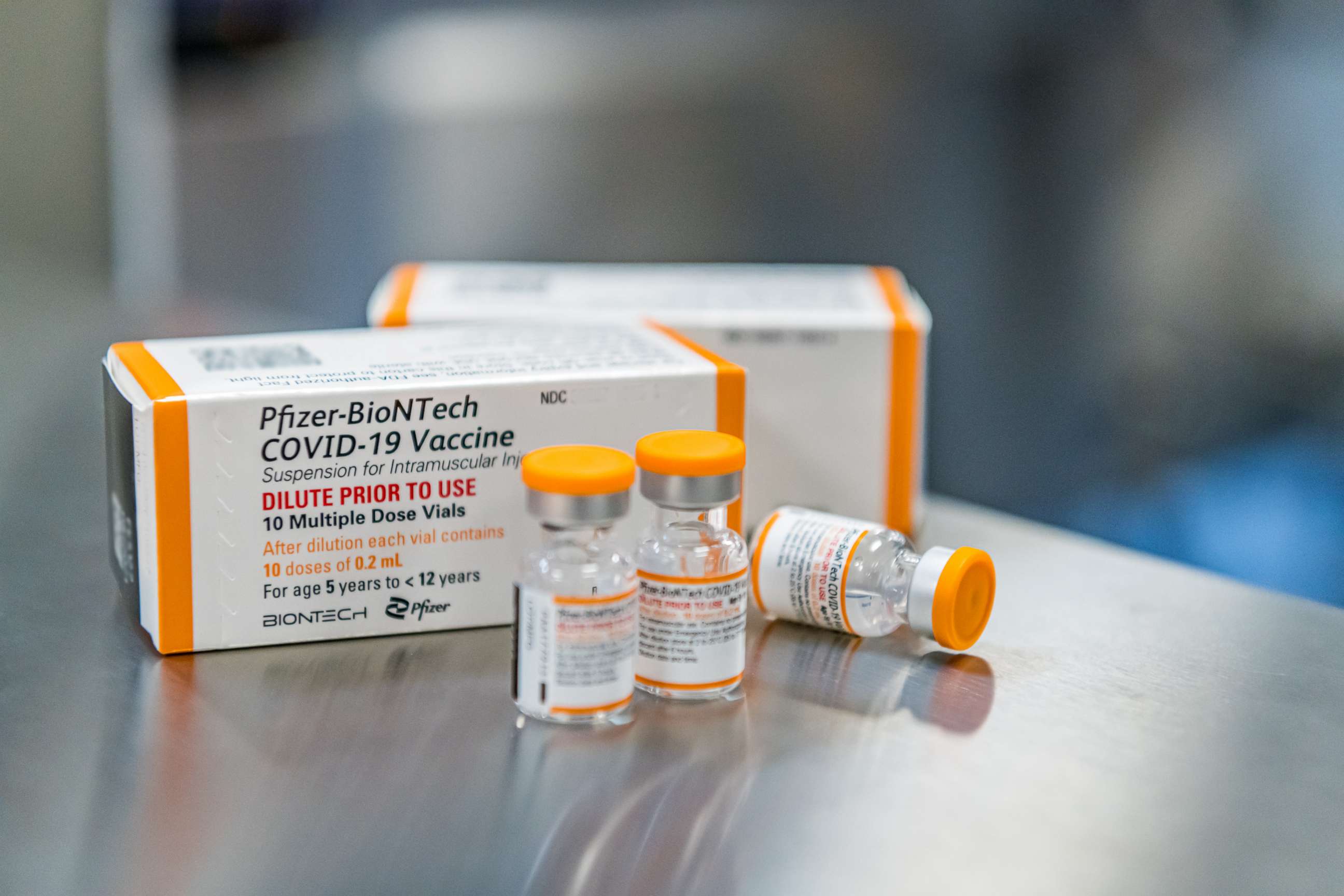
Differing immune systems among people of different ages also help explain why the cutoffs for vaccine eligibility rest on age and not body size.
In addition to the COVID-19 vaccine, other immunizations are also scheduled and administered based on age and not weight. This is partially due to the fact that the body’s immune responses to vaccinations and infection are known to be different based on age.
3. Kids can get both flu shots and COVID-19 shots.
Like adults, children can receive the flu shot at the same time as the COVID-19 shot, according to the CDC.
Children under 12 , like adults, should also get vaccinated against COVID-19 even if they have already had the virus, according to the CDC.
The agency is also reminding parents not to give kids a pain reliever before the vaccination to try to mitigate side effects.
Instead, parents are urged to talk to their doctor about a non-aspirin pain reliever after the shot if needed.
4. The FDA will decide later on full approval for the vaccine.
Following normal protocol, the FDA will continue to review data to decide whether to grant full FDA approval for the vaccine in kids ages 5 to 11.
The FDA approved the Pfizer vaccine for people ages 16 and older in August. It is currently authorized for emergency use in children ages 12 to 15.

The two other vaccines currently available in the U.S., Moderna and Johnson & Johnson, are currently available only for people 18 years and older.
Moderna announced in late October that it plans to submit data to the FDA soon showing its vaccine for children ages 6 to 11 produces a strong immune response and appears safe.
5. Families need to remain vigilant against COVID-19.
Unvaccinated children can not only become ill from COVID-19 themselves, but they can also spread the virus to more vulnerable family members and other adults with whom they interact.
Both the CDC and the American Academy of Pediatrics recommend universal mask-wearing in schools to help slow the spread of COVID-19.
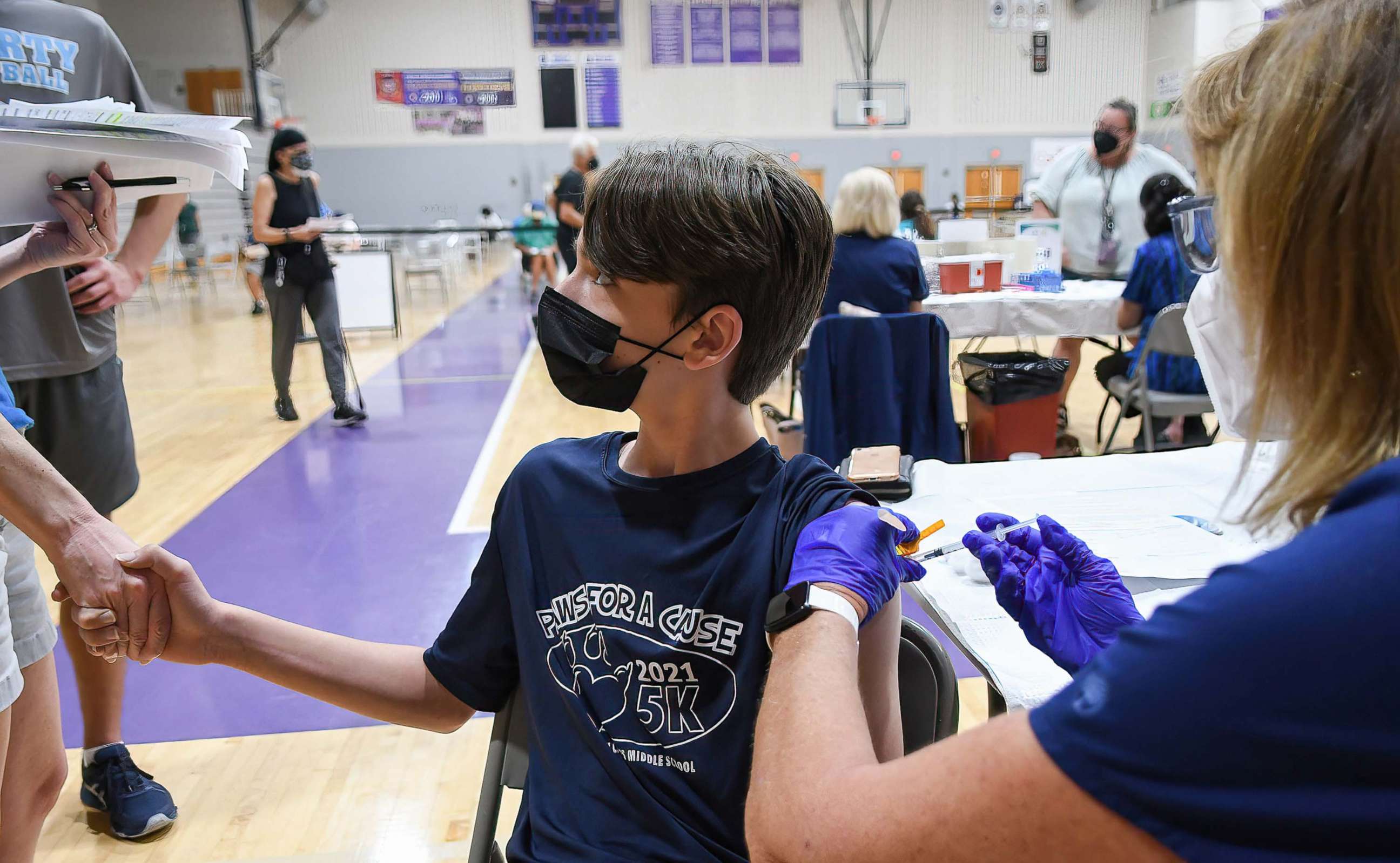
Experts said that in addition to unvaccinated children wearing face masks, parents and siblings who are vaccinated should also continue to wear face masks indoors because of the rates of breakthrough infections in the U.S.
Families should also continue to follow other safety guidelines shared throughout the pandemic, including social distancing and hand-washing.
ABC News' Sasha Pezenik and Anne Flaherty contributed to this report.