Parents cautious of new FDA-approved device for kids with ADHD
The newly approved device is being met with “cautious optimism” by parents.
The U.S. Food and Drug Administration’s approval of the first medical device of its kind to treat ADHD in children between the ages of 7 and 12 years-old is being met with "cautious optimism" by parents and advocates.
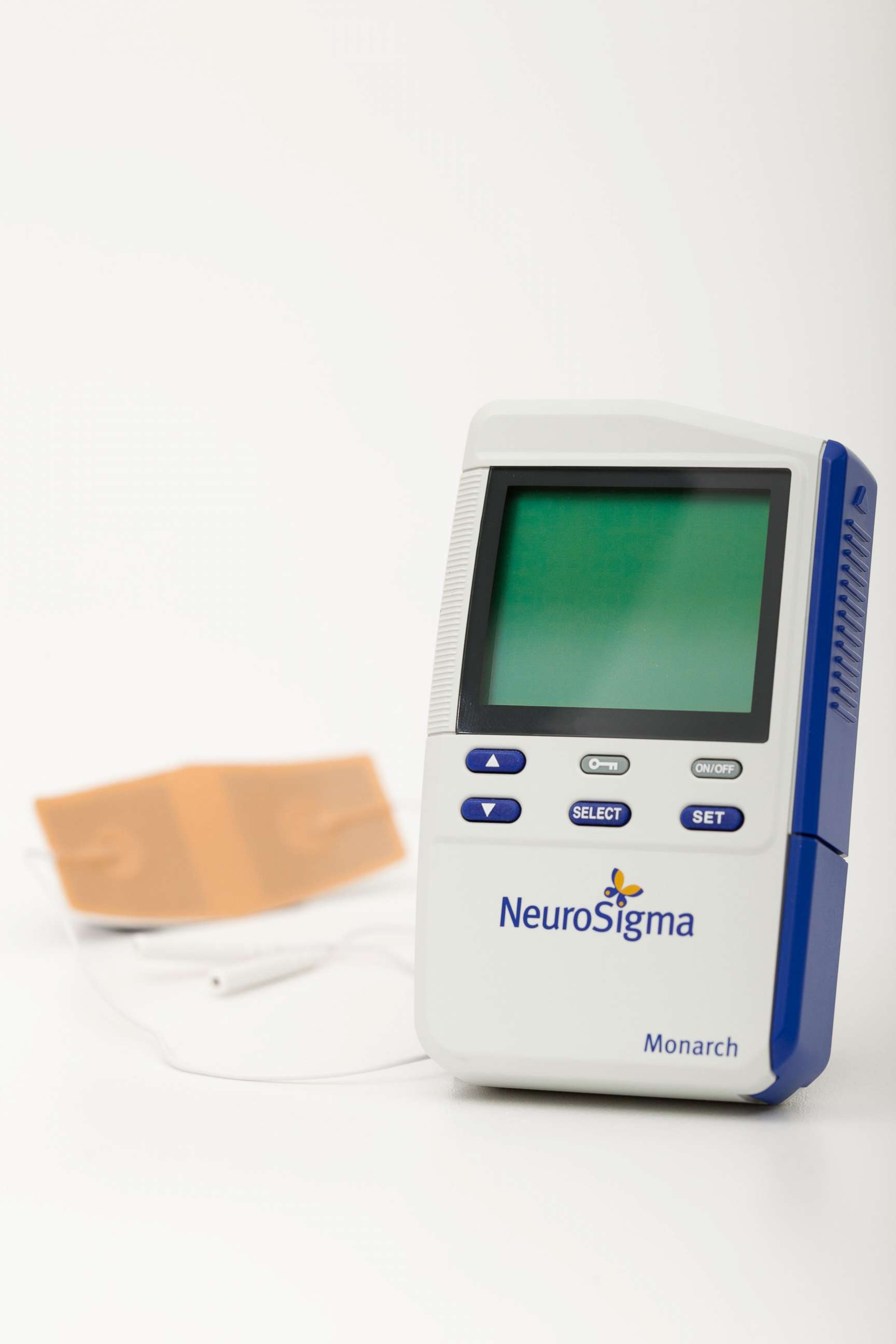
The new device known as the Monarch eTNS system will cost parents about $1,000 and isn’t yet covered by insurance. It’s the size of a cellphone and works by attaching a small patch to a patient’s forehead. It delivers a low-level stimulation to the parts of the brain thought to be involved in ADHD.
"This new device offers a safe, non-drug option for treatment of ADHD in pediatric patients through the use of mild nerve stimulation, a first of its kind," said Carlos Peña who heads up the FDA’s division of neurological and physical medicine devices.
In a clinical trial done over four weeks involving 62 children with moderate to severe ADHD, there was "statistically significant improvement" in reducing their symptoms. But some doctors are cautiously optimistic, urging more research before parents start buying these devices.
"The study draws upon a very small sample size," said Dr. Max Wiznitzer, co-chair of the professional advisory board for Children & Adults with ADHD. "Most of the known ADHD drugs—Ritalin, Adderall—have a far larger sample size and so one study is just not enough to cite efficacy."
But the known side of effects of those drugs includes stomach ache, decreased appetite nervousness and sleeping problems.
The new Monarch eTNS device, however, is not without its own side effects including drowsiness, headaches, and an increased appetite.
"It’s probably going to be a good treatment for kids with moderate ADHD symptoms," said Dr. Wiznitzer. "But my question is whether this device alone will be sufficient."
Dr. Andy Leuchter, director of the Transcranial Magnetic Stimulation Clinical and Research program at UCLA, was involved in the research phase of the device’s clinical trial. Dr. Leuchter believes the Monarch eTNS system is something parents can trust, but also says more research, including potentially pairing it with other known ADHD medication, would help.
"This is as close to a side effect free treatment device that you’re going to get," he said. "So there’s very little downside risk to seeing if there’s a benefit."
But even for parents who are struggling to help their children respond to the effects of their own ADHD medication, the device presents a level of uneasiness.
Andrea Frank is a mom to two teenage boys in Lodi, Wisconsin, who were both diagnosed with ADHD when they were four-years-old, but have now chosen to take themselves off of treatment. She started a virtual support group for parents of children with ADHD, which has more than 40,000 members worldwide.
"I personally would not go that way," she said. "I see a lot of frustrated parents who feel they’re hitting a wall but because it’s an electric based device, there’s just a fear among parents to use this."




