How COVID-19 vaccine rollout compares to smallpox, polio and others in the past
What we can learn from the pitfalls and successes of past programs.
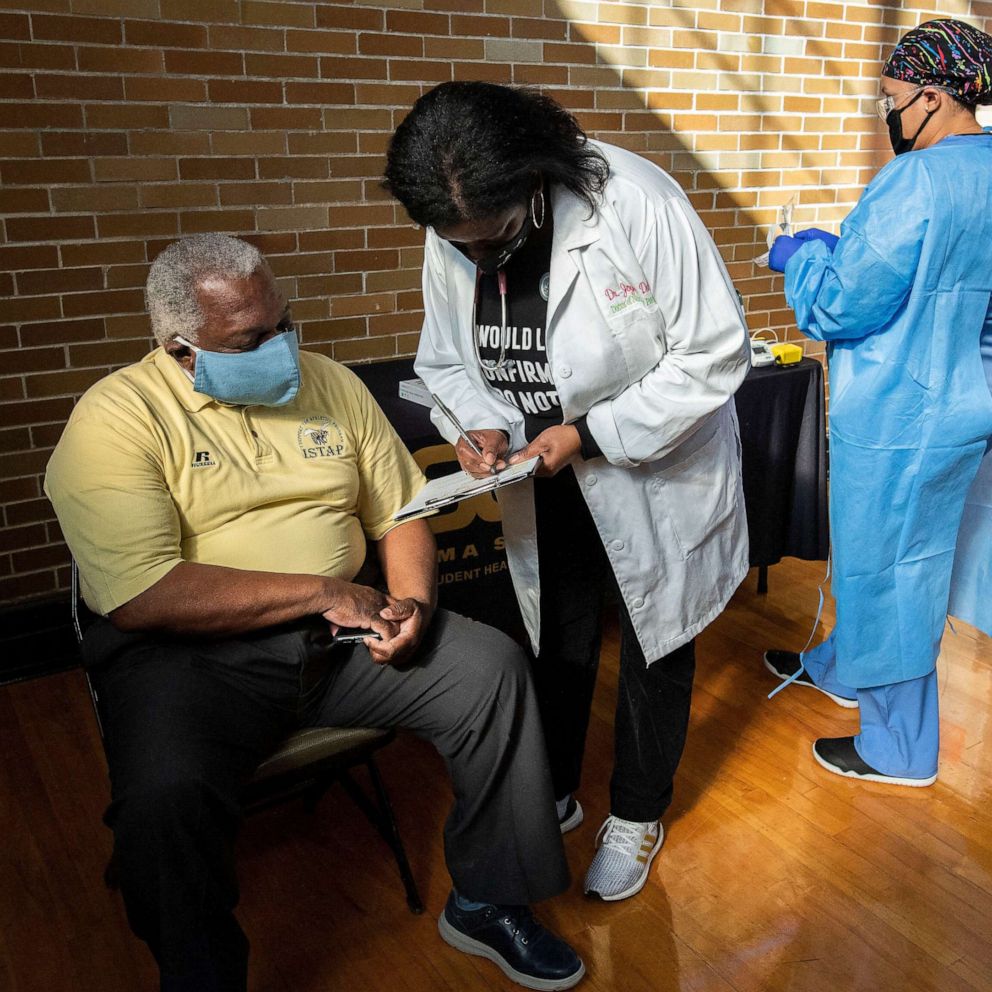
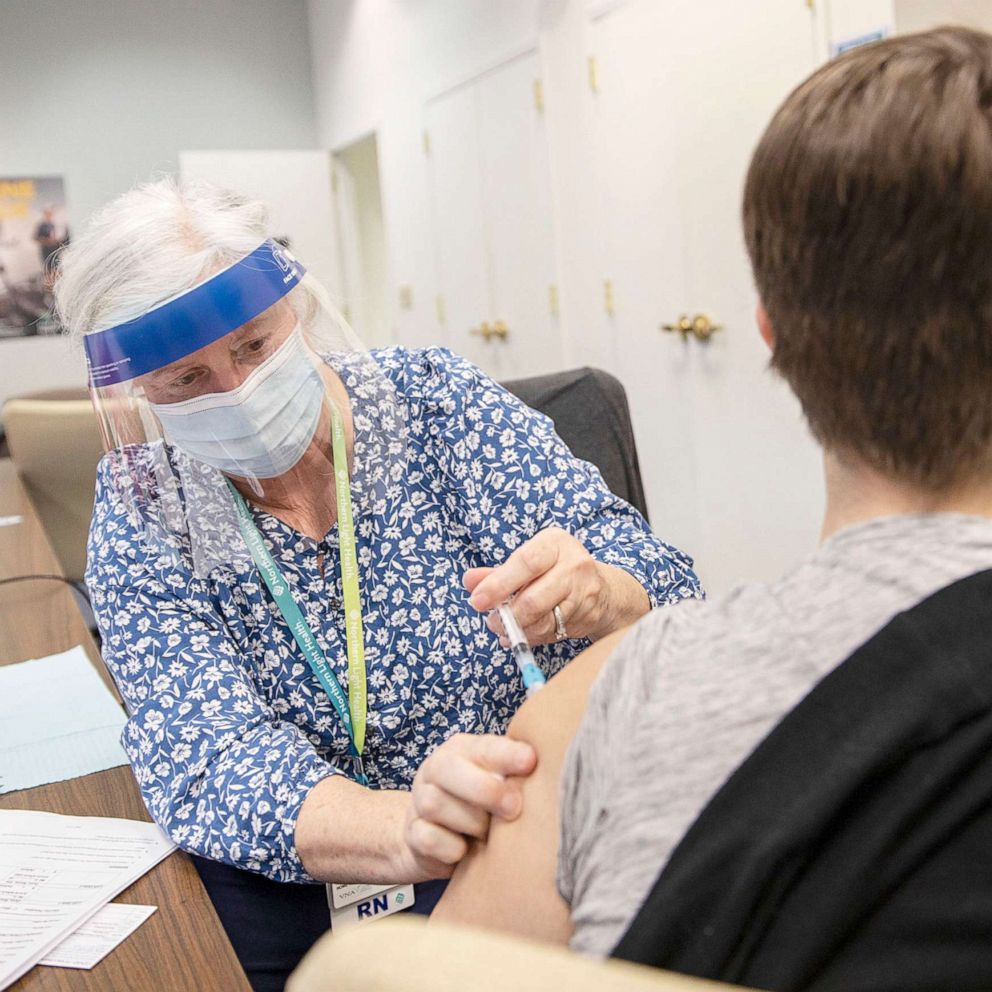
When President Joe Biden took office, he inherited several highly effective COVID-19 vaccines and hope for ending the pandemic, thanks to the record pace of development and novel technology.
But the vaccine rollout has been another story, some say -- a distribution process that had fallen short of the Trump administration's goal of administering 100 million doses by the end of 2020 -- and that critics say has left the country in a race against time as new and more contagious variants of the virus take hold.
"While the vaccine provides so much hope, the rollout has been a dismal failure thus far," Biden said. "I understand the despair and frustration of so many Americans and how they're feeling."
By early February, the United States had distributed more than 49 million vaccine doses, with 26 million people receiving one or more doses, according to the Centers for Disease Control and Prevention. But the program itself, which spans two administrations, has been plagued by lack of supply, staffing issues and communication failures between states and the federal government.
On occasion, the fragile vaccines, one of which needs to be stored at ultra-cold temperatures, have expired on hospital shelves. In others instances, vaccination appointment no-shows and vaccine hesitancy have created a free-for-all situations where health workers scramble to offload expiring doses to people who want them. And some have criticized Biden's pledge of 100 million shots in 100 days as being too slow while countries like Israel, for instance, are making much more progress.

So how do other mass vaccination efforts at previous points in American history compare?
Rene Najara, an epidemiologist and editor of the History of Vaccines project at the College of Physicians of Philadelphia, said that the COVID-19 vaccine rollout is difficult to compare to historical U.S. vaccine drives. There are a confluence of obstacles hindering the current effort, including politics, social media, mass travel and vaccine hesitancy, that are trickier than in years past, he explained, while the scope of the effort -- vaccinating everyone -- is wider.
"Given all the circumstances surrounding this pandemic, it's gone as well as it can," Najara said. "Can it be better? Absolutely. But would require something other than vaccine strategy," he said, like a less divided country or a solution for the racial inequality that's underpinned every aspect of the U.S. pandemic response, from testing to treatment and now vaccination.
With those caveats in mind, examining the pitfalls and successes of past vaccine drive can be instructive. "Hopefully we will fight to retain the memory and lessons from this, so that we can avert the next pandemic or make it a bit more tolerable," Najara said. "Because we will have another one."
What we can learn from our past vaccination programs:
Smallpox, 1947: The city-wide success story
After a man who traveled by bus from Mexico City to New York City in 1947 was hospitalized and died of smallpox, the city embarked on a vaccination spree so successful that it's now the stuff of infectious disease lore.
A vaccine for smallpox had been developed roughly 150 years prior, so when a second person died of smallpox, the mayor urged all 7.8 million New Yorkers to get vaccinated and mobilized police, fire, health departments and hospitals to make space for vaccinations. The resulting rollout was swift. Despite long lines, the city estimates that roughly 6 million New Yorkers were vaccinated in six weeks.

In the end, only 12 people were sickened and two died. "There are situations that mirror what we're seeing, but nothing like [COVID-19]," Najara said of New York's smallpox campaign. "We're trying to vaccinate the whole planet. It's quite the undertaking. You can seen why there are delays," he added.
Polio, 1955: Before eradication, a tragic mistake
While it's not a perfect comparison to the COVID-19 vaccine program, "the closest thing in my experience is the polio vaccine rollout," Najara said.
Polio, a contagious disease that can cause nerve damage, paralysis and death, and which mainly affects children younger than 5, terrified parents.
"You had an emergency situation, because it was affecting children," Najara said. Unlike today, vaccine hesitancy wasn't a widespread deterrent, so when an effective vaccine against polio was announced in 1955, "almost every single parent in the U.S. mobilized to get their child vaccinated," he said.
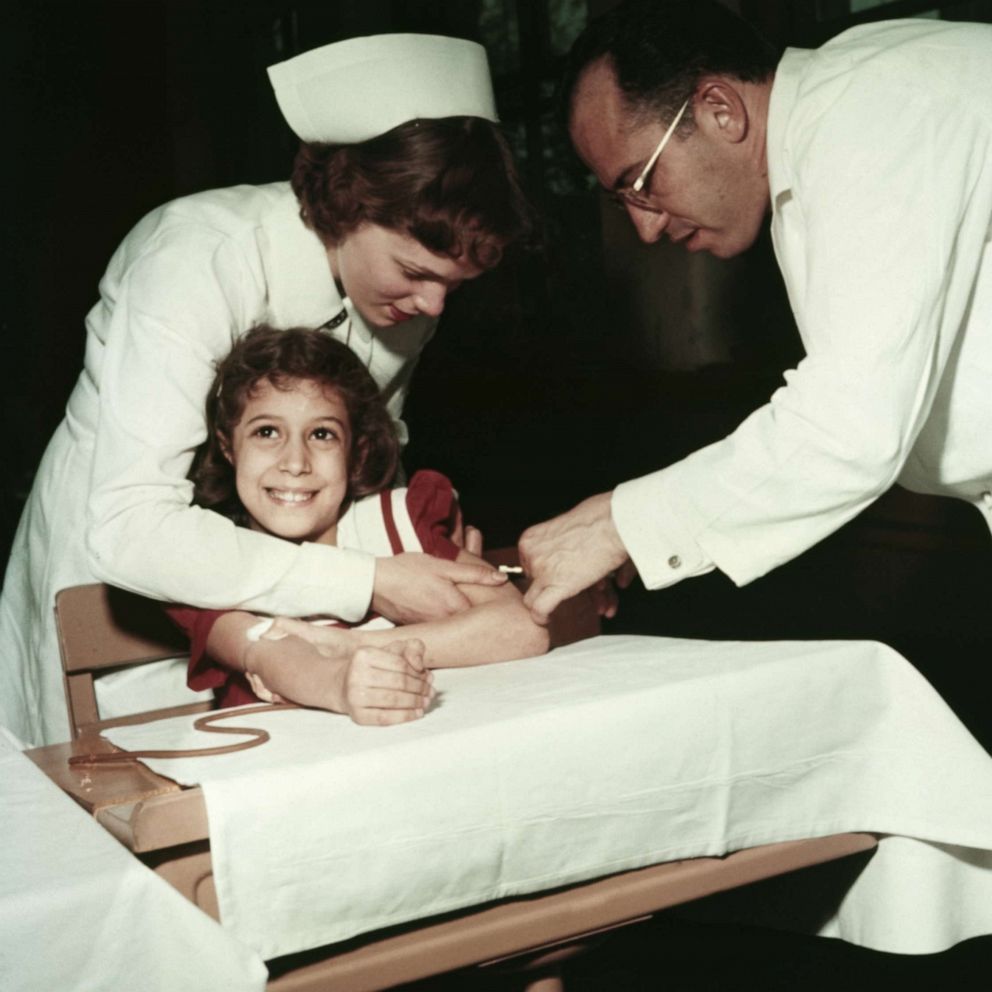
But not long after the rollout began, a manufacturing error at Cutter Laboratories in California led to a bad batch of vaccines. When 11 people died and hundreds were paralyzed, the surgeon general temporarily suspended the program.
"The Cutter Incident was a defining moment in the history of vaccine manufacturing and government oversight of vaccines," according to the CDC. The incident led to the passage federal vaccine safety regulations and polio vaccinations started again in the fall of 1955.
The renewed effort worked. Polio cases fell from 35,000 in 1954 to 5,500 cases in 1957, according to the CDC. Today, the U.S. is polio-free, with the last naturally occurring case of polio occurring in 1979, according to the Mayo Clinic.
Swine flu, 1976: The fake-out
Following an outbreak of swine flu at a New Jersey military base in 1976, President Gerald Ford announced a plan to vaccinate everyone in the country against the disease.

Similar to the vaccine rollout for COVID-19, the federal government provided vaccines to state health departments and to private medical practices. Although the program managed to get shots in 40 million Americans' arms during the course of a year, Ford's campaign involved fast-tracking a vaccine during an election year.
When no pandemic came to pass and 30 people out of the 40 million vaccinated died from adverse reactions to the vaccine, the CDC's credibility suffered.
H1N1, 2009: Business as usual
One advantage for scientists responding to the H1N1 pandemic was that H1N1 was a novel influenza virus. "We were used to making flu vaccines," Najara said.
The first U.S. infection was detected in April and by September there was a vaccine to fight it. Unlike the Pfizer and Moderna vaccines for COVID-19, which require two dose and cold storage, vaccinating for H1N1 didn't require special logistics.
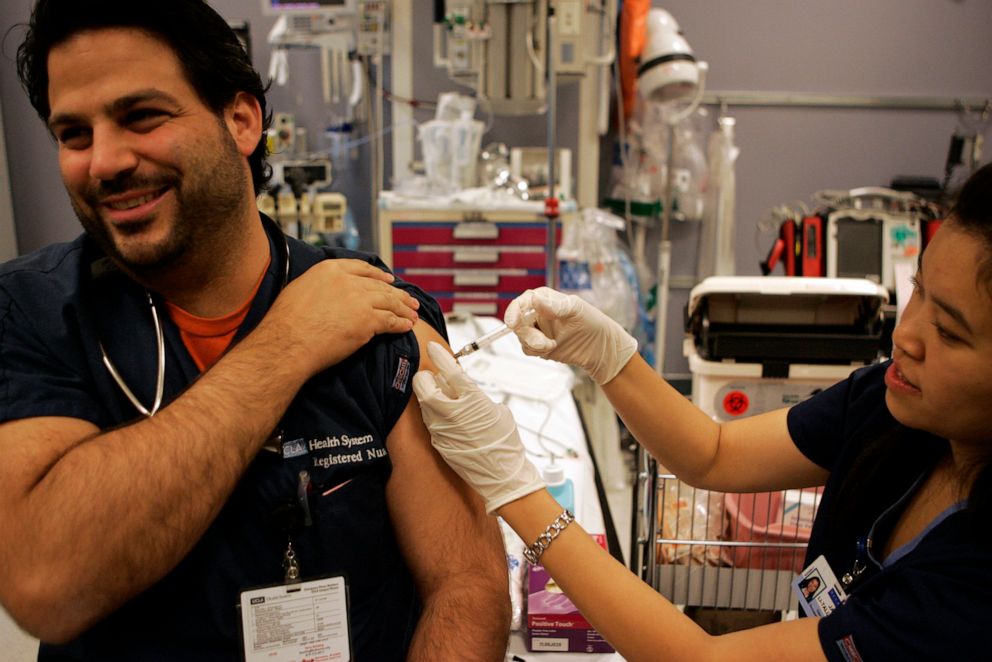
"It was a regular vaccine rollout of the flu vaccine," Najara said. "Everything was in place."
By August 2010, less than two years after it began, the World Health Organization had declared an end to the H1N1 influenza pandemic, although H1N1 continues to circulate as a seasonal flu virus, according to the CDC. In total the U.S. reported 60.8 million cases and 12,469 deaths during the course of the outbreak.
What to know about the coronavirus:
- How it started and how to protect yourself: Coronavirus explained
- What to do if you have symptoms: Coronavirus symptoms
- Tracking the spread in the U.S. and worldwide: Coronavirus map
Tune into ABC at 1 p.m. ET and ABC News Live at 4 p.m. ET every weekday for special coverage of the novel coronavirus with the full ABC News team, including the latest news, context and analysis.