FDA authorizes faster, cheaper COVID test in push to pioneer progress
SalivaDirect is a "testing innovation game changer," Adm. Brett Giroir said.
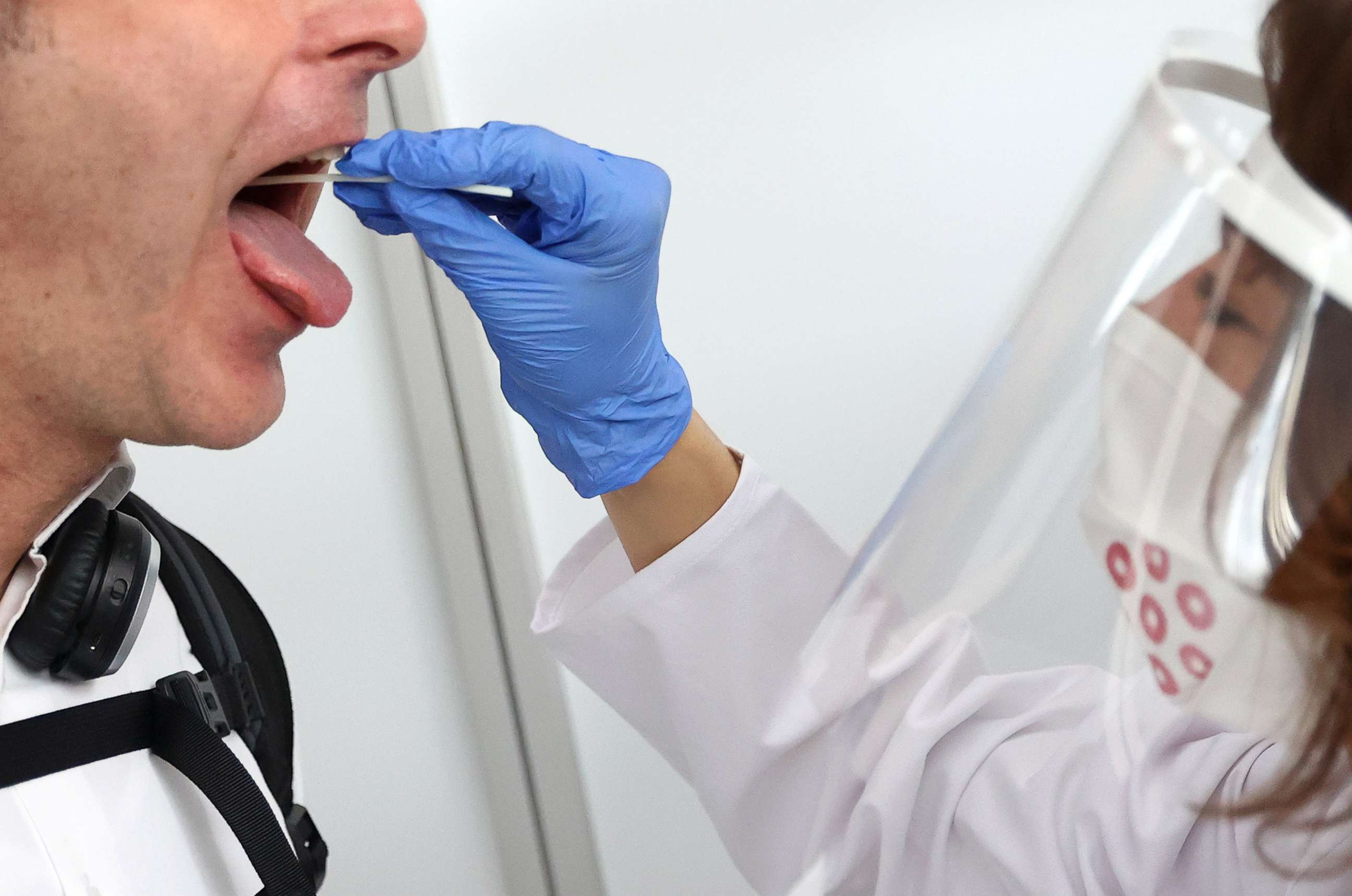
In what could be a major breakthrough for novel coronavirus testing, the Food and Drug Administration has given emergency authorization for a new kind of saliva-based screening method Saturday, aimed at streamlining how samples are processed and cutting both cost and turnaround time.
Developed by Yale's School of Public Health, the SalivaDirect test offers an approach unique in its attempt to bypass some of the more time-consuming and cost-prohibitive supplies and aspects of the testing process.
Although the FDA has authorized other saliva tests, SalivaDirect marks the first RNA extraction-free method authorized for emergency use, skipping one of the more expensive and labor-intensive steps: pulling out the virus's genetic material. Instead, the Yale method breaks saliva down directly with an enzyme and applies heat.
It doesn't require Spectrum collections tubes or certain chemical reagents, which can be expensive and in scarce supply.
Yale aims to have the total cost of their tests run about $10 -- an enormous departure from other similar tests, like the one developed by Rutgers' lab, which has also partnered with Vault Health, for a kit that can cost $150.
If it works, and works well, it could greatly expand testing capacity at a lower price point, what the government's testing czar, Adm. Brett Giroir, called a "testing innovation game changer."
"This, I hope, is a turning point," Dr. Anne Wyllie, associate research scientist at the Yale School of Public Health and one of the leads on SalivaDirect, told ABC News. "Expand testing capacity, inspire creativity and we can take competition to those labs charging a lot, and bring prices down. And that we can show people what can be done."
Yale is now in talks with labs in both academic and commercial spheres, Wyllie told ABC News. Among them, Jackson Lab in Connecticut, Quest and Vault Health. Their test is self-collected under supervision, opening the door for further telehealth opportunities.
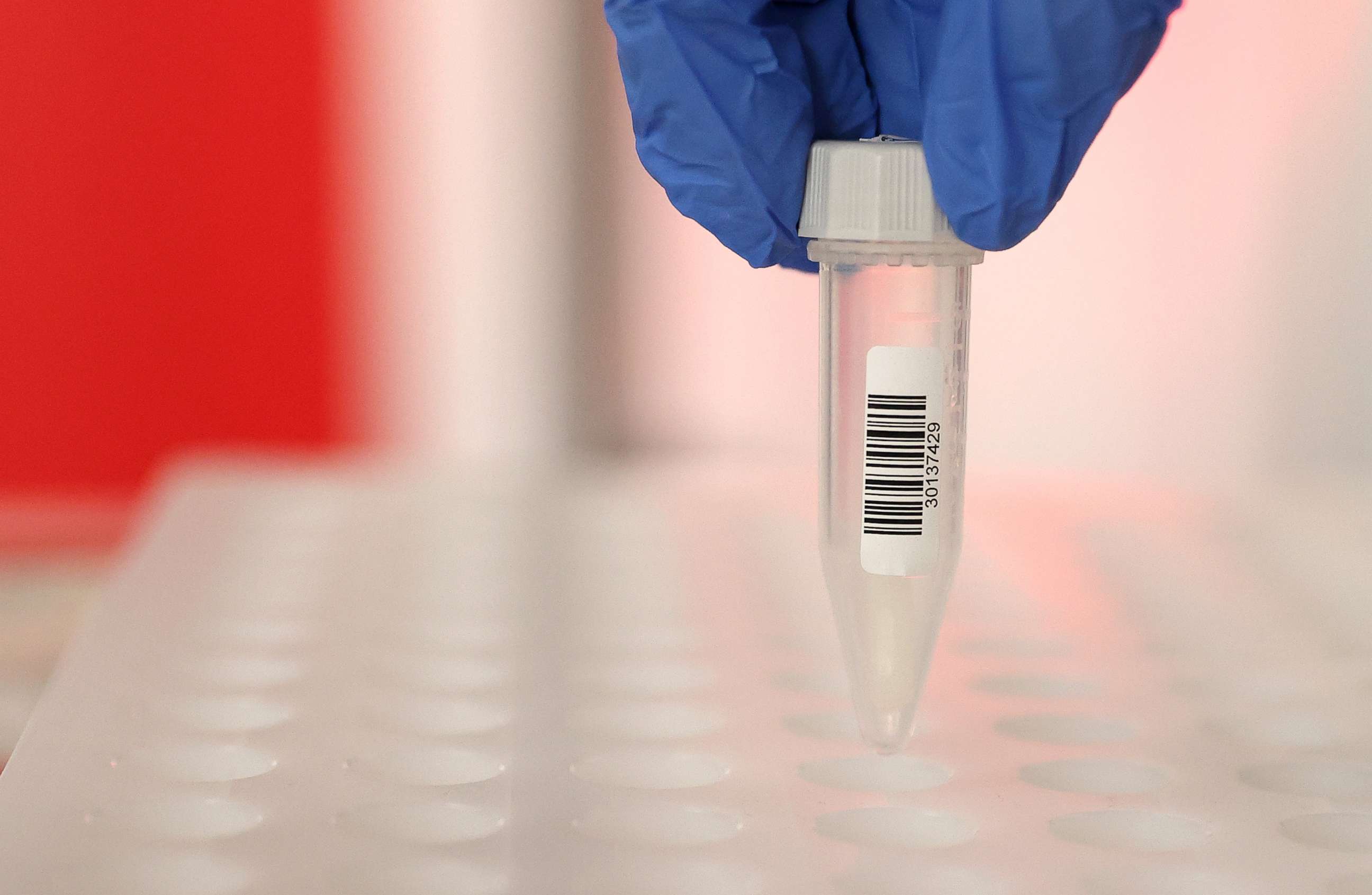
Wyllie told ABC News that their venture yields no profit for them. Their method will be offered as an open-source protocol to laboratories across the country to throttle up coronavirus testing.
Moving forward, Wyllie said, they seek to expand their research into the realm of asymptomatic screening. Even while waiting on their emergency authorization, their research team has been working with the NBA, with opt-in screening for players, coaches and staff -- and their study has been jointly funded by the NBA and NBA Players' Association. The NBA is also a client of Vault Health via the Rutgers test; among the several screening methods they use within and without their bubble, thus far, they have reported no confirmed positive cases.
Questions still remain as to whether saliva testing is as sensitive or effective as nasal swabs; they've been eyed as a less-invasive method that also helps mitigate the deluge of demand and results bottleneck with which major labs have had to contend.
In very limited studies, saliva tests have shown results that match nasal swab tests almost exactly. But the small sample size -- just 60 cases -- used to authorize the Rutgers lab test has left some skepticism.

Wyllie knows the margin for error is slim, especially in offering a test so attractive for universities looking to keep a stringent screening process in place for their thousands of students.
"Our work isn't over," Wyllie said. "We just want there to be more and more affordable options. This is just another step. But we have to keep moving forward."
ABC News' Sony Salzman, Stephanie Ebbs, Brian Hartman and Eric Strauss contributed to this report.



