FDA authorizes 1st COVID-19 'breathalyzer' test
Results are detected within just a few minutes, the agency said.
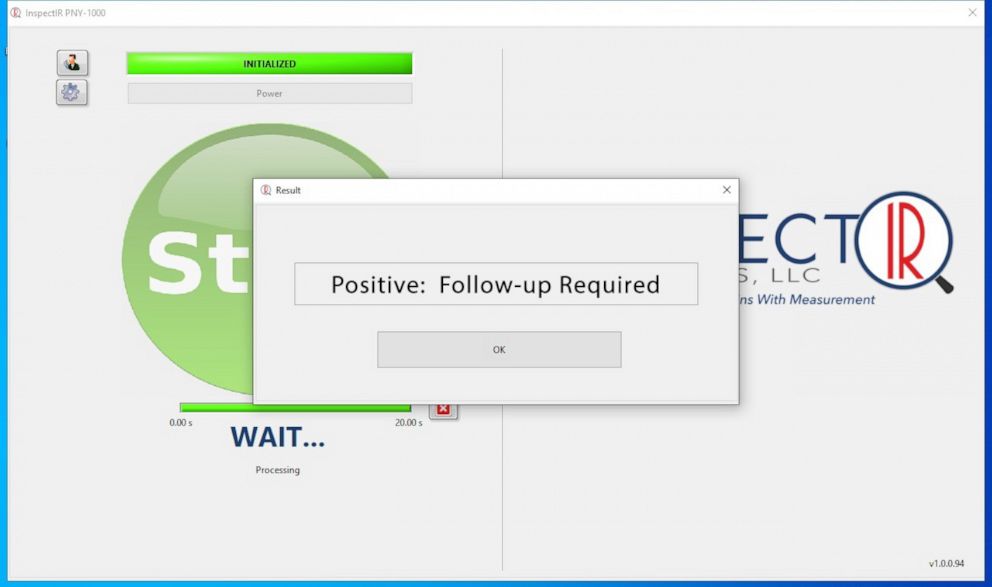
The Food and Drug Administration has granted emergency use authorization for a COVID-19 test that uses breath samples -- the first of its kind to get the agency's green light. The device, called the InspectIR Covid-19 Breathalyzer, is "about the size of a piece of carry-on luggage," and can accurately detect coronavirus on the breath within just a few minutes, the company and FDA said.
While other COVID-19 testing methods have used nasal swab or saliva samples to detect viral particles, this test uses a technique called gas chromatography-mass spectrometry to separate and identify chemical mixtures and rapidly screen for five compounds associated with a COVID-19 infection in a person's exhaled breath.
A positive result with this device is still considered "presumptive," however, and should still be confirmed with a PCR test, the so-called "gold standard" in COVID-19 screening, the FDA said. The agency warned negative results "should be considered in the context of a patient's recent exposures, history and the presence of clinical signs and symptoms consistent with COVID-19, as they do not rule out SARS-CoV-2 infection and should not be used as the sole basis for treatment or patient management decisions."
While it comes with caveats, the company views its product as a potential game-changer in the large-scale COVID-19 screening arena.
"We spent a lot of time and a lot of effort on the science and the technology," company co-founder Luke Kaiser said. "We are very focused on having a great product and a true product that can go anywhere, and test accurately."
While this test offers rapid results -- promised in under three minutes -- this is not the same kind of rapid test available for purchase at local pharmacies. It is not aimed at being an "at-home" screening method -- rather, it is meant for what InspectIR Systems COO John Redmond described to ABC News as a "volume play."
InspectIR Systems aims to produce roughly 100 test devices per week, with 10 made so far, Kaiser told ABC News. In the next month, they expect to have roughly 250 test devices ready to go and say they will be making "as many as the line can hold."
The company anticipates leasing test devices to companies and within industries ideal for en masse screening, as would be appropriate within the health care industry, such as nursing homes, prisons and the travel and hospitality industry, such as cruise lines, and perhaps schools. Redmond said the company expects leasing agreements to cost between $25,000 and $30,000 per month, which is why this would be most appropriate for that "volume play" setting.
Though that dollar figure sounds large, the idea is to get the cost per test down to an average of $10 to $12 each, Redmond said, which is in line with and perhaps even cheaper than commercially available at-home rapid tests. Baked into that leasing price would be a supply of individually wrapped paper straws, an air filter for the test kit and other necessary components.
With a single-use sanitary paper straw people blow their breath sample, about the amount it takes "to inflate a small balloon," into the system, the company said, which looks for the chemistry and compounds associated with COVID-19.
The test must be done with supervision from a health care professional at doctor's offices, hospitals, mobile testing sites or other venues with qualified staff on hand.
Each device can each be used to evaluate approximately 160 samples per day. At this level of production, testing capacity using the InspectIR COVID-19 Breathalyzer is expected to increase by approximately 64,000 samples per month.
"Today's authorization is yet another example of the rapid innovation occurring with diagnostic tests for COVID-19," Dr. Jeff Shuren, director of the FDA's Center for Devices and Radiological Health, said in a statement. "The FDA continues to support the development of novel COVID-19 tests with the goal of advancing technologies that can help address the current pandemic and better position the U.S. for the next public health emergency."
ABC's Dave Packer reports:
ABC News' Eric M. Strauss contributed to this report.




