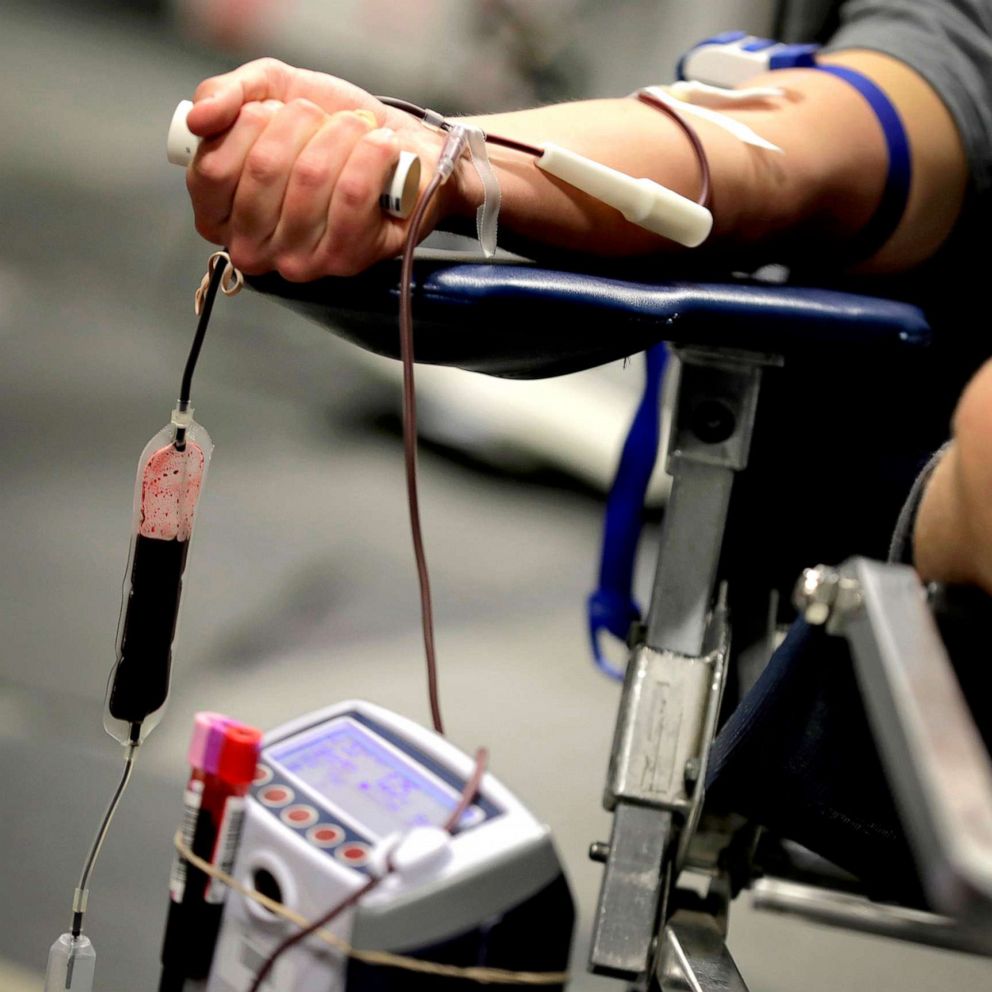
FDA loosens restrictions on gay and bisexual men, encourages blood donations amid coronavirus crisis
Officials encouraged people to donate during the coronavirus emergency.
The Food and Drug Administration on Thursday said it would loosen some of the restrictions that have blocked gay and bisexual men from donating blood.
The agency is changing the recommended deferral period for men who have had sex with another man from 12 months to three months. Restrictions on blood donations from gay and bisexual men, and other groups considered to be high risk for HIV or AIDS transmission, date back to the 1980's.
Surgeon General Jerome Adams called the FDA decision "tremendous" and potentially life-saving at a time when overall blood donations have fallen and hospitals face critical shortages as people stay home and blood drives are canceled because of the coronavirus pandemic.
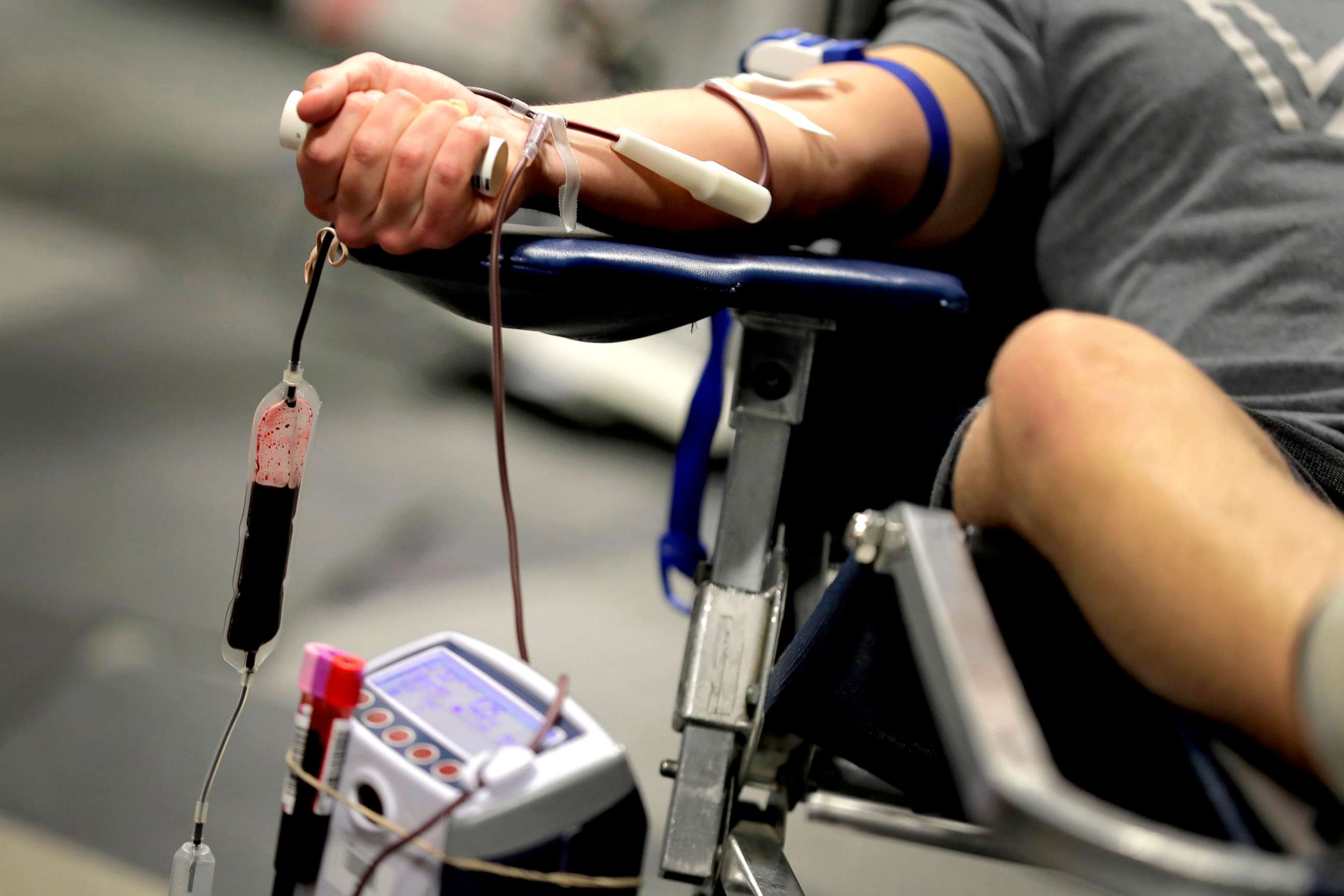
"It is critically important we have rationally and scientifically based blood donation deferral periods," Adams said on a call with reporters. "In particular, we know that reducing the deferral period for men who have sex with men can significantly increase life-saving blood donations, prevent drug shortages and help reduce harmful stigma experienced by the MSM community."
Tune into ABC at 1 p.m. ET and ABC News Live at 4 p.m. ET every weekday for special coverage of the novel coronavirus with the full ABC News team, including the latest news, context and analysis.
The FDA said the changes would be implemented immediately and and are expected to remain in place after the COVID-19 pandemic ends.
The revised guidance also includes people with recent tattoos and piercings and women who have had sex with a gay or bisexual man; the recommended deferral period is now three months versus 12 months.
Peter Marks, director of the FDA's Center for Biologics Evaluation and Research, said in a statement: "Based on recently completed studies and epidemiologic data, we've concluded that the current policies regarding the eligibility of certain donors can be modified without compromising the safety of the blood supply."
Marks said tens of thousands of people could now be eligible to donate for the first time and organizations that previously objected to the referral policy may be more willing to host blood drives.
The Human Rights Campaign, the largest LGBTQ civil rights organization, recognized the FDA’s move as a step forward but said there’s more work to be done.
"While this change by the FDA is a step in the right direction, it still bases itself in bias rather than science," Alphonso David, president of the Human Rights Campaign, said in a statement. "This is progress from the FDA. But our work is not yet done."
David said it was "critical to prioritize science and facts over fear and bias," referring to the current COVID-19 pandemic.
He continued, "As the global pandemic wears on, the integrity and safety of the blood supply in this country must be preserved, strengthened, and maintained. Continuing to enforce the de facto prohibition on blood donation by sexually active gay and bisexual men does not reflect the best science available. We must right this wrong now and without delay."
In the last week Democratic senators and gay rights advocacy groups have called on the federal government to loosen these restrictions, citing the recent blood shortages caused by the COVID-19 outbreak.
"LGBTQ Americans can hold their heads up today and know that our voices will always triumph over discrimination," GLAAD President and CEO Sarah Kate Ellis said in a statement. "This is a victory for all of us who raised our collective voices against the discriminatory ban on gay and bisexual men donating blood. The FDA’s decision to lower the deferral period on men who have sex with men from 12 months to 3 months is a step towards being more in line with science, but remains imperfect. We will keep fighting until the deferral period is lifted and gay and bi men, and all LGBTQ people, are treated equal to others."
The American Red Cross in November recommended an update to the current policies.
"As a scientifically-based interim step, the Red Cross encourages the Food and Drug Administration (FDA) to consider reducing its deferral time for men who have sex with men from twelve to three months while further options are evaluated for the United States," the organization said at the time.
What to know about coronavirus:
- How it started and how to protect yourself: coronavirus explained
- What to do if you have symptoms: coronavirus symptoms
- Tracking the spread in the US and Worldwide: coronavirus map
Editor's note: This was originally published on April. 2, 2020.